Where to Find an Air Density Calculator Excel Spreadsheet
To find an air density calculator Excel spreadsheet to use as an air density calculator, click here to visit our spreadsheet store. Why use an online calculator or look in tables, when you can get an air density calculator excel spreadsheet to use as an air density calculator here? Read on for information about Excel spreadsheets that can be used to calculate the density of air (and other gases) at different pressures and temperatures with the ideal gas law.
Gas density background for an Air Density Calculator Excel Spreadsheet
Pressure and temperature have significant effects on the density of gases, so some means of determining the density of air and other gases at specified temperatures and pressures is needed for a variety of fluid mechanics applications. Fortunately, the ideal gas law provides a means of doing this for many gases over ranges of temperature and pressure that are of interest.
The Ideal Gas Law for use in an Air Density Calculator Excel Spreadsheet
A common form for the ideal gas law equation is PV = nRT, giving the relationship among T, the absolute temperature of the gas; P, its absolute pressure; V, the volume occupied by n moles of the gas; and R, the ideal gas law constant.
The density of the gas can be introduced into this equation, through the fact that molecular weight (MW) has units of mass/mole, so that n = m/MW. This leads to the ideal gas law written as: PV = (m/MW)RT. Solving this equation for m/V (which is equal to the gas density, ρ) gives the following equation for gas density as a function of its MW, pressure and temperature: ρ = (MW)P/RT.
A commonly used set of U.S. units for this equation is as follows:
ρ = density of the gas in slugs/ft3,
MW = molecular weight of the gas in slugs/slugmole (or kg/kgmole, etc.) (NOTE: MW of air = 29),
P = absolute gas pressure in psia (NOTE: Absolute pressure equals pressure measured by a guage plus atmospheric pressure.),
T = absolute temperature of the gas in oR (NOTE: oR = oF + 459.67)
R = ideal gas constant in psia-ft3/slugmole-oR.
For conditions under which air can be treated as an ideal gas (see the next section), the ideal gas law in this form can be used to calculate the density of air at different pressures and temperatures.
The air density calculator excel spreadsheet template shown in the screenshot below will calculate the density of a gas for specified molecular weight, pressure and temperature. This Excel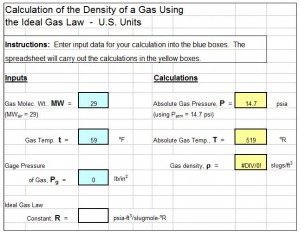 spreadsheet is available at a very reasonable price in our spreadsheet store and can be used with either U.S. or S.I. units. These spreadsheets also contain tables of critical temperature and critical pressure for several common gases.
spreadsheet is available at a very reasonable price in our spreadsheet store and can be used with either U.S. or S.I. units. These spreadsheets also contain tables of critical temperature and critical pressure for several common gases.
But When Can I Use the Ideal Gas Law to Calculate the Density of Air?
A good question indeed, because air and other gases for which you may need a density value are real gases, not ideal gases. It is fortunate, however, that many real gases behave almost exactly like an ideal gas over a wide range of temperatures and pressures. The 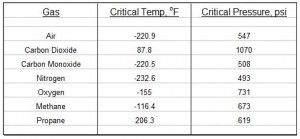 ideal gas law works best for high temperatures (relative to the critical temperature of the gas) and low pressures (relative to the critical pressure of the gas). See table at the left for values of critical temperature and critical pressure for several common gases. For many practical, real situations, the ideal gas law gives quite accurate values for the density of air (and many other gases) at different pressures and temperatures.
ideal gas law works best for high temperatures (relative to the critical temperature of the gas) and low pressures (relative to the critical pressure of the gas). See table at the left for values of critical temperature and critical pressure for several common gases. For many practical, real situations, the ideal gas law gives quite accurate values for the density of air (and many other gases) at different pressures and temperatures.
S.I. Units for the Ideal Gas Law
The ideal gas law is a dimensionally consistent equation, so it can be used with any consistent set of units. For SI units the ideal gas law parameters are as follows:
ρ = density in kg/m3,
P = absolute gas pressure in pascals (N/m2),
T = absolute temperature in oK (NOTE: oK = oC + 273.15)
R = ideal gas constant in Joules/kgmole-K
References:
1. Bengtson, Harlan H., Flow Measurement in Pipes and Ducts, An online PDH course for Professional Engineers
2. Munson, B. R., Young, D. F., & Okiishi, T. H., Fundamentals of Fluid Mechanics, 4th Ed., New York: John Wiley and Sons, Inc, 2002.
3. Applied Thermodynamics ebook, http://www.taftan.com/thermodynamics/
4. Bengtson, Harlan H., “Gas Property Calculator Spreadsheet,” an Amazon Kindle e-book.
Pingback: Gas Compressibility Factor Calculator Excel Spreadsheet